Diphenyl phosphorazidate (DPPA)
CAS 26386-88-9, Cat. No EN300-55951
Reagent for stereospecific conversion of alcohols to azides, Curtius rearrangement
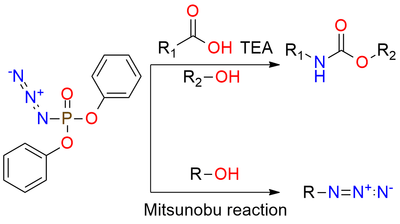
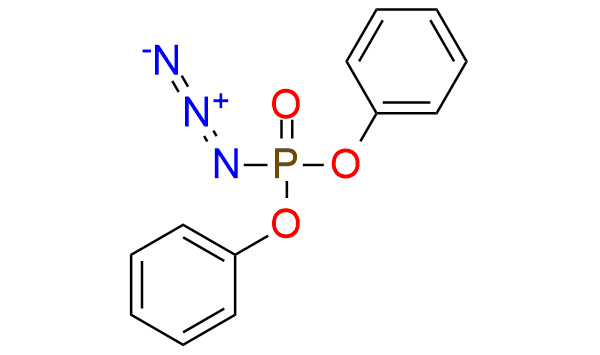
DPPA is a useful reagent that is stable azide and can be utilized for the conversion of alcohols to azides via the Mitsunobu reaction or for Curtius rearrangement. It is a shelf-stable colorless liquid, that is soluble in toluene, THF, DMF, and t-butyl alcohol. It also can be used for the synthesis of α-aryl carboxylic acids. Alkyl aryl ketones are converted into the corresponding α aryl alkanoic acids via a three-step sequence. This method offers good yields, exhibits high functional specificity, and uses less toxic reagents[1].
Synonyms: O,O-diphenylphosphoryl azide; [Azido(phenoxy)phosphoryl]oxybenzene; Azidodiphenoxyoxophosphorane; Diphenyl azidophosphate
Selected publication
1. Diphenyl Phosphorazidate.
Thomas A. V.; Ghosh A. K.; Sridhar P. R. Encyclopedia of Reagents for Organic Synthesis 2008. DOI: 10.1002/047084289X.rd434.pub2
